Chapter 12: Fungal sex and fungal groups

The ‘vegetative’ (i.e., not associated with sex and reproduction) structure of most fungi are generally very consistent: they are composed of branched filaments that explore the volume of material that they feed on (be it living or dead) and this structure provides for acquisition of materials that they need to grow and survive. The consistency of vegetative form makes it difficult to classify fungi because they mostly look alike, although some vary on the basis of whether or not the filaments on coenocytic or septate (with cross walls and therefore cellular). However, fungi do show variation, and can be grouped, based upon structures that are associated with sexual reproduction. The exceptions to this are the two groups where sex is either completely absent (Glomeromycota — the endomycorhizal forming fungi) or rare (Chytridomycota — the chytrids). The remaining three groups (bread molds = Zygomycota, cup fungi = Ascomycota, and club fungi = Basidiomycota usually exhibit specific features associated with sexual reproduction that are used define these groups (generally considered phyla). Fungi that show no sexual features used to be put into a group called the ‘Fungi Imperfecti’ (Deuteromycota), but molecular techniques now allow these fungi to be placed in one of the groups mentioned above.
Fungi have the following features associated with sex and reproduction that are different from most familiar sexual organisms:
- Reproduction often involves both sexual aspects and asexual aspects. Cells called spores — defined as cells with both dispersal capabilities (i.e., mobile cells) and reproductive capabilities (i.e., cells that can grow into new fungal organisms) are often important in both asexual and sexual reproduction.
- Gametes (cells that can fuse with other gametes) are quite different from what most would consider ‘typical’; they are usually hyphal cells (part of the fungal filaments) that have the ability to fuse with other hyphae. Depending upon the group, these special hyphae may or may not have structural features that would distinguish them from the normal hyphae.
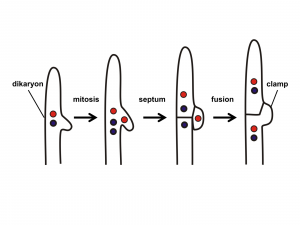
- Syngamy often involves two steps separated in time and often in space. This is a consequence of the fact that the fusion of the hyphae serving as gametes is generally not followed immediately by the fusion of nuclei. Cellular fusion is called plasmogamy and nuclear fusion is called karyogamy. Plasmogamy followed by a delayed karyogamy allows fungi to have a novel condition, the dikaryon state, where a cell has two nuclei (‘dikaryon’ means ‘two nuclei’), one from each parent. This condition is often perpetuated: the dikaryon cell divides while both nuclei divide, thereby forming a new cell that is also dikaryon. This process can continue, producing multiple dikaryon cells and dikaryon hyphae. Note that although a dikaryon cell has two copies of each chromosome, it is not considered diploid because each nucleus is haploid, with only one copy of each chromosome.
- At some point, some of the dikaryon cells become diploid as a result of the fusion of the two nuclei (karyogamy). The diploid cells are NOT perpetuated; they undergo meiosis to form haploid nuclei which develop walls to become haploid cells. Thus, the diploid state is very brief in extent (i.e., size and number of cells) and often brief in time as well — the diploid cells never divide mitotically to form more diploid cells, they only divide meiotically to form haploid cells. These haploid cells are, or soon become, spores that are dispersed from the parent fungus to a new location where they germinate and form haploid hyphae.
- ‘Mating strains’ are a common mechanism that ensures that fungi do not mate with themselves. Within a particular species there are two to many mating types. A particular mating type needs to find a different mating type in order to interact sexually (i.e., fuse hyphae). The simplest situation has two mating types: “+” and “-” and they need to find each other to mate, but there may be more than just two types, e.g., types A, B, C, D, E. And mating type A could mate with any of the others but not with another A. Generally, the interaction, or lack of it, involves chemical signals (pheromones) that are emitted by one mating type and sensed only by fungi of a different mating type. In response to the pheromone, hyphae grow and find each other, bringing about plasmogamy.
TOPICS
-
- Zygomycetes (bread molds)
- Ascomycetes (cup fungi)
- Basidiomycetes (club fungi)
Zygomycetes, the bread molds
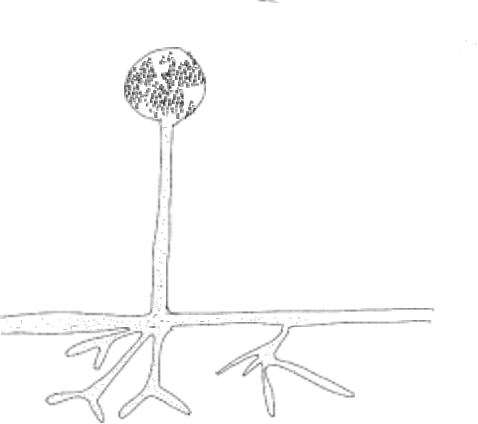

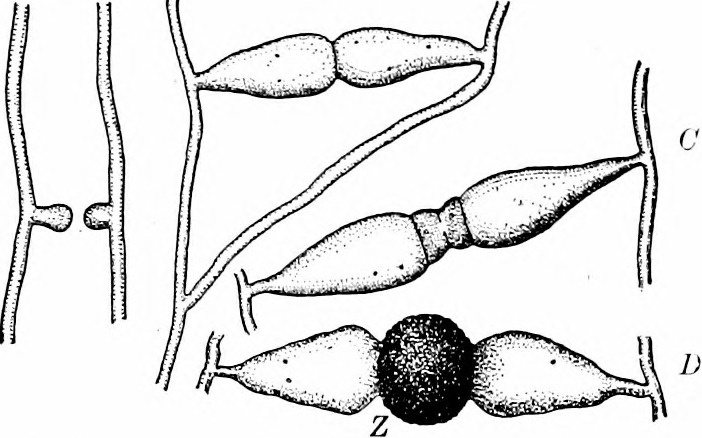
In the zygomycetes (Fig. 2-3), most reproduction is asexual and results from the production of a stalked structure (sporangiophore) terminated with spherical sporangium. Inside the sporangium is the only cellular tissue produced by the group, meaning it is a structure where walls are present and individual nuclei are packaged one to a cell. These cells mature into spores that are dispersed when the sporangium disintegrates. Much less frequent is reproduction associated with a sexual process when a specific structure, the zygospore, is formed and from which the group gets its name. The process is initiated when two compatible hyphae sense each other’s presence because of pheromones. This triggers hyphae grow towards each other with the production of specialized hyphal branches that are capable of fusing to each other. Since bread molds are coenocytic, the fusion of two hyphae (plasmogamy) allows multiple nuclei to come into proximity (Fig 4). Unusual for the group, two cross walls, one in each of the fusing hyphae, form near the point of fusion, creating a single coenocytic cell with nuclei derived from each of the two mating strains (Fig. 5-6). This is the only dikaryon cell produced by the bread molds and it develops into a structure called a zygospore (Fig. 7-8) by enlarging slightly and developing a thick wall. Inside the zygospore haploid nuclei of one type pair with nuclei of the other type and fuse (karyogamy) to form diploid nuclei, transforming the cell from being dikaryon to being diploid, but it is still multinucleate. The zygospore is generally dormant and generally has more specific germination requirements and a longer lifespan than the asexual spores. When the zygospore germinates it produces a sporangiophore comparable to those produced asexually with a sporangium at its tip. As this develops the diploid nuclei undergo meiosis and the haploid nuclei are individually packaged into spores as cell walls are produced. The spores are subsequently dispersed, and when they germinate they produce haploid, coenocytic hyphae. Note that the dikaryon state is limited to a single cell and has a very brief existence.
- Fig. 4 When hyphae of different mating strains are in proximity, pheromones trigger hyphal branches of each strain to grow towards each other.
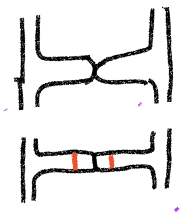
- Fig. 5 When the two strains make contact, new cell walls (in red) form, producing two multinucleate cells at the tip of each branch other.
- Fig. 6 The cell walls where the two hyphae made contact are broken down, plasmogamy occurs, creating a multinucleate cell that is dikaryon, with haploid nuclei from the two parents present. The haploid nuclei of the two types find each other, fuse and thus form a coenocytic cell with diploid nuclei (plus some unmatched haploid nuclei). At the same time the cell develops into a zygospore with a thick cell wall. It matures and becomes dormant.
- Fig. 7 When the zygospores germinate the diploid nuclei undergo meiosis and the zygospore produces a stalk with a sporangium containing haploid cells; these are dispersed.
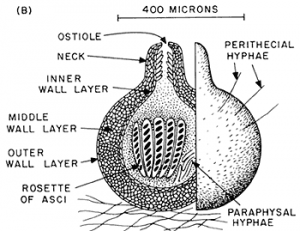
Ascomycota — the cup fungi

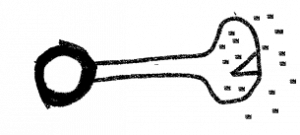
Unlike the bread molds, the cup fungi (Ascomycetes), are septate, i.e., they have cross walls and the ‘feeding hyphae’, the ones that acquire nutrients, are constructed of cells that have a single haploid nucleus. Many members of the group, reproduction is primarily to exclusively asexual, a consequence of the production of specialized hyphal branches that produce small cells (conidia) that are easily broken off to serve as propagules. Generally, these cells are dormant and have other cellular features, e.g., stored food, that promote their role in reproduction and dispersal. When and if sexual reproduction occurs, it is initiated when compatible mating strains are close to each other and communicate by pheromones. Inflated hyphal branches are produced, one on each strain, producing cells that are slightly different in form and are multinucleate. When they are close to each other one of the cells produces an extension that touches the expanded cell of the other mating type and fuses with it (plasmogamy occurs) and haploid nuclei move from one of the structures, termed the antheridium and considered male, to the other one, called an ascogonium and considered female. As a result of the migration of nuclei, the archegonium cell becomes dikaryon, with two types of nuclei, one from each parent. The ascogonium develops extensions and two nuclei (one from each mating type) associate and migrate into them. A cross wall is formed, producing the first cell of what is described as an ascogenous hypha. These hyphae grow from the tip and remain dikaryon, with two haploid nuclei per cell, a result of a coordination of tip expansion, two nuclear divisions (one for each nucleus), nucleus migration, and septum formation. Growth of the ascogneous hyphae plus growth of both (haploid) parental hyphae produce a fruiting body of densely intertwined hyphae. The size of the fruiting body ranges tremendously in size, from roughly 100 um to 10 cm or more, with most at the smaller end. The size and shape vary and can be used to identify species. The common name for the group (‘cup fungi’) relates to a cup shaped fruiting body. Cup fungi are found both living independently and also as the fungal partner of a number of lichens; many lichens produce ascocarps (Fig. 10) that are relatively large and visible to the naked eye. Other fruiting bodies are flask-shaped or completely closed and typically are less than a mm in extent.

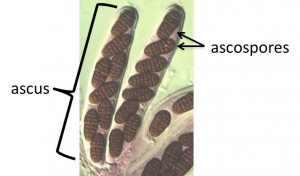
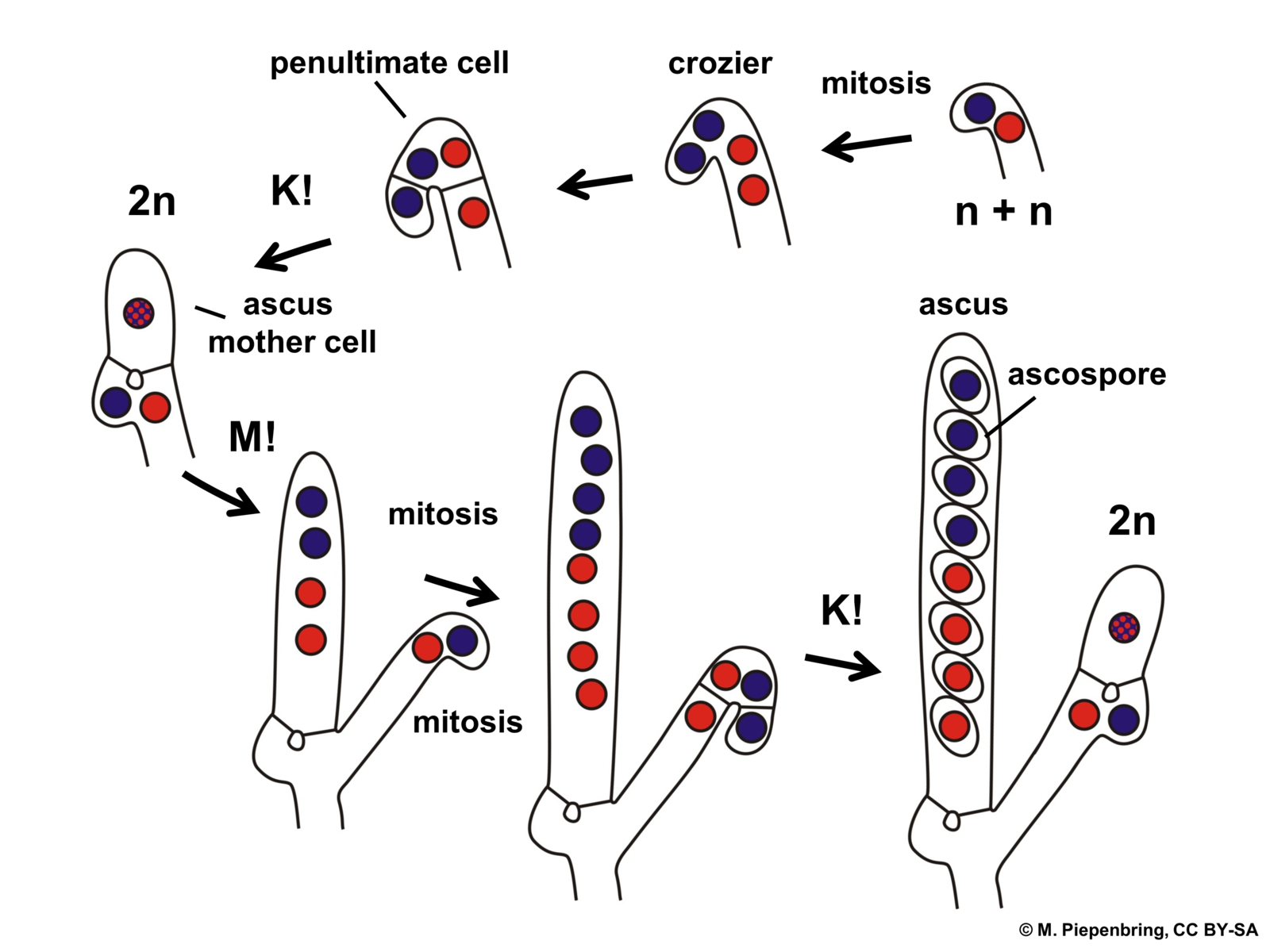
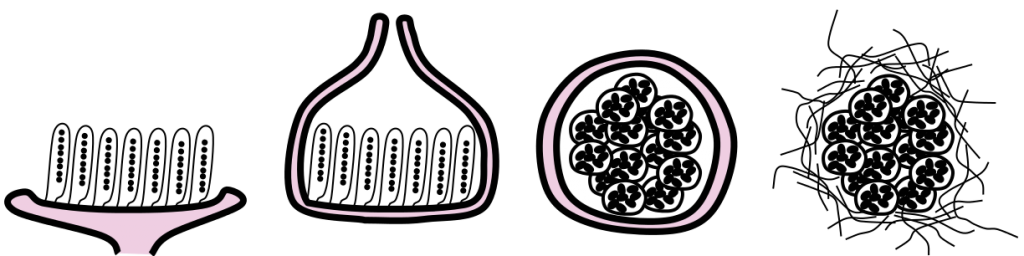
In a specific part of this fruiting body the sexual cycle is completed. The tips of the ascogenous hyphae form a hook, turning back on itself and the two nuclei fuse (karyogamy occurs) in the cell that has formed the hook (crozier), making it (briefly) a diploid cell. The only diploid cells found in the Ascomycota are these specific cells, eventually called asci (singular = ascus) situated at the ends of the ascogenous hyphae. These hyphae produce no additional cells after forming the hook. The diploid cell elongates and then undergoes meiosis to form an elongate cell with four haploid nuclei. Each of these then go through mitosis to produce an elongate cell called an ascus with eight haploid nuclei in a row. Each nucleus acquires a cell wall and develop into what is called an ascospore. Asci with eight ascospores are diagnostic for the Ascomycota.
Hydrostatic pressure within the ascus causes ascospores to be forcibly dispersed when the tip of the ascus ruptures. Dispersed ascospores germinate to form haploid hyphae, which form haploid mycelia which may reproduce asexually via conidia or other spores. If a haploid mycelium comes in contact with hyphae of a different mating type, sexual reproduction may be triggered.
- Fig. 12 The ascus—a characteristic feature of the Ascomycota
- Fig. 13 Note size, many ascocarps (ascomycete fruiting bodies) are quite small


Basidiomycetes—club fungi
The club fungi are septate like the cup fungi (Ascomycota). Of all the fungal groups the club fungi have the most extensive dikaryon state. As is the case for almost all fungi, the dikaryon state is initiated when two compatible haploid, monokaryon hyphae find each other as a result of chemical attraction. Plasmogamy occurs, forming a dikaryon cell. This cell divides and grows extensively, forming a feeding mycelium that is dikaryon. Many of the basidiomycota produce ‘clamp connections’ (Fig. 17) that may help to maintain the dikaryon state as the hyphae elongate. The nuclei undergo mitosis, and cross walls formed. As the terminal cell elongates an arch is formed between the terminal cell and its parental cell. When the two nuclei divide the arch allows one of the two nuclei to move to the parental cell as septa form. This ensures that both the daughter cell and mother cell has one of each type of nucleus.
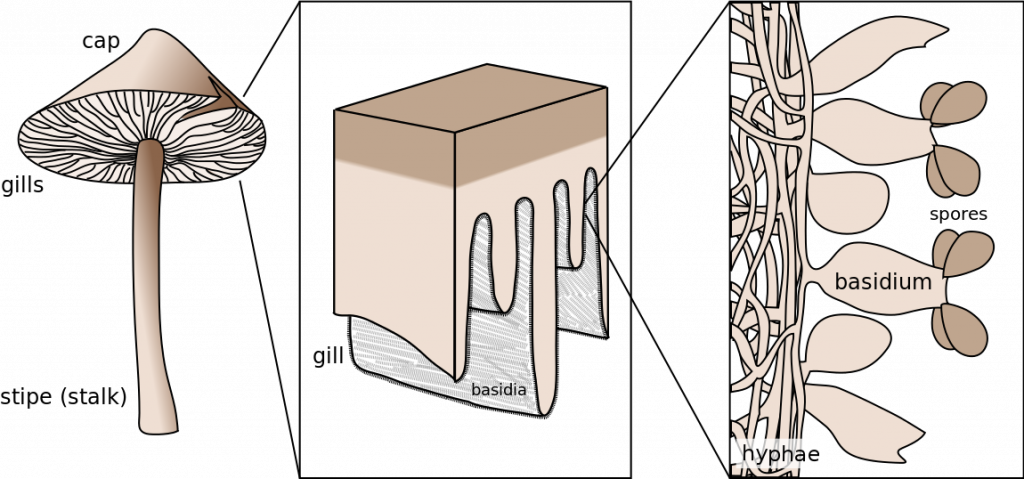
Recall that that there is only a single dikaryon cell in the bread molds (Zygomycota) and the only dikaryon cells of the the cup fungi (Ascomycota) are found in the fruiting bod ies. Thus, if one encounters a dikaryotic hyphae outside of a fruiting body then it must be long to a club fungus (Basidiomycota). The dikaryotic hyphae of club fungi grow and feed extensively until appropriate conditions are encountered to trigger ‘fruiting’. At this point the growth pattern of at least some of the hyphae changes from one where their substrate is explored for nutrition to one where hyphae intertwine with each other, forming the dense mass of hyphae that will become fruiting body. Often the fruiting body emerges from the substrate that the fungus is feeding on, e.g., a mushroom emerging from the soil or from the trunk of a tree. Somewhere on or in the dense mass of hyphae special cells are produced, termed basidia, that are ‘club-shaped’ (Fig. 18). Inside these cells karyogamy occurs, transforming them into diploid cells. The diploid nucleus undergoes meiosis to produce four haploid nuclei, but no cytokinesis occurs, making the basidium ‘quadra-nucleate’. Four extensions grow out of the basidium and the four nuclei migrate into these. This is followed by cell wall formation to produce a basidium with no nucleus but with four loosely attached haploid cells, called basidiospores, extending off from them. The spores are ballistically released and are dispersed by the wind. When they germinate they form new haploid hyphae.
Unlike the cup fungi, whose fruiting bodies are generally small and not typically noticed, the fruiting bodies of many club fungi are often relatively large, 10-30 cm and have characteristic shapes that we describe as mushrooms (with a stalk and a cap) (Fig. 19-20) or bracket fungi (roughly hoof shaped and attached without a stalk to the trunks of woody plants). Two very common patterns of basidia distribution are (1) extending from thin fins of tissue termed ‘gills’, typical of the commercially available mushroom, or (2) basidia extending from tubes (cylinders) of tissue which, when viewed at the surface, appear as numerous pores.





Two large groups of Basidiomycetes, the smuts (Fig. 22-23) (Class Ustomycetes) and the rusts (Fig. 24-25) (class Teliomycetes) have slightly different and sometimes involved patterns of sex and reproduction associated with their obligate parasitic lifestyle. Many of these organisms are very important economically because they can drastically reduce yields of important crops. Neither form ‘fruiting bodies’, made solely of fungal material, but rather cause abnormal growth (galls) on the plant that they are growing in. These galls are composed of infected plant cells, uninfected plants cells growing abnormally because of the parasite, and some fungal hyphae interconnecting infected cells. While the typical basidiomycete (described above) has a single type of spore, the haploid basidiospores formed in the fruiting body, both rusts and smuts commonly produce teliospores, consisting of one to several dikaryotic cells. When teliospores germinate karyogamy occurs, followed by meiosis, producing a basidium with haploid nuclei that produce haploid basidiospores.

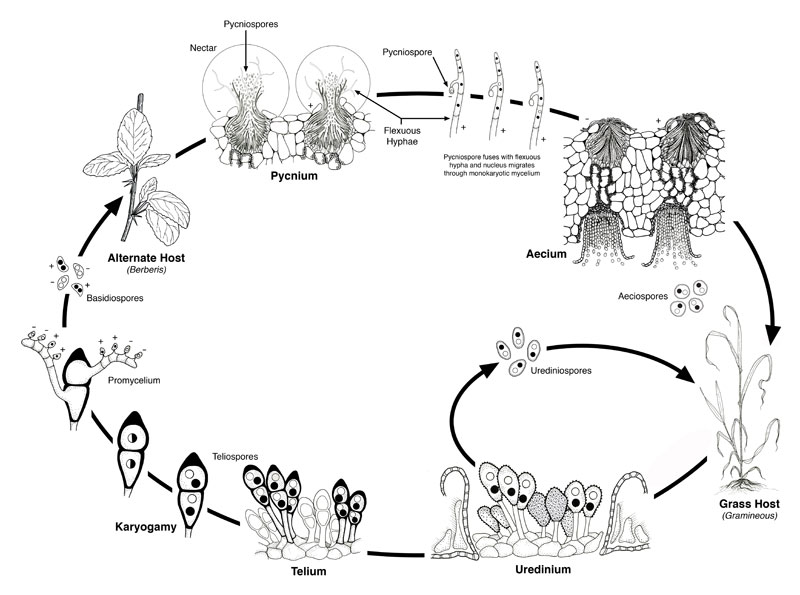
While most smuts only produce these two types of spores, rusts may have two or three more types of spores and a complicated life cycle that sometimes involves two hosts. An example of a rust with two hosts is discussed below (Fig. 26) and also in Chapter 30.

Basidiospores infect host #1 (barberry) and form structure called pycnia on the upper surface of the leaf. Pycnia produce two structures that bring about syngamy. One of these produces haploid spores called pyncospores (or spermatia) that might be considered gametes. Pycnospores are released as single cells into a sweet ‘nectar’ that attracts insects who can transport them to other compatible (i.e., different mating type) pycni a where they can fuse (plasmogamy) with the second structure involved in syngamy that is produced by pycnia: ‘flexuous hyphae’ that extend out of the pycnia. Fusion of the pyncospore with the haploid cells of these hyphae brings about syngamy and forms the initial cell of a dikaryon hyphae. This grows to the bottom of the leaf, forms a cup shaped structure that releases dikaryon spores (aeciospores) that infect host #2 (wheat), causing it to form another type of dikaryon spore, uridinospores, that can infect more wheat plants. Late in the season the final type of spore, the teliospore, forms on wheat. Karyogamy and meiosis occur in teliospores and from them emerge promycelia (basically basidia) that produce basidiospores.
Media Attributions
- Cantharellus pallens © zaca is licensed under a CC BY-SA (Attribution ShareAlike) license
- Bread mold growing on a tomato © Comrade King is licensed under a CC BY-SA (Attribution ShareAlike) license
- Rhizopus zygospore © Leavitt, Robert Greenleaf, Gray, Asa is licensed under a Public Domain license
- Peziza spec. © James Lindsey is licensed under a CC BY-SA (Attribution ShareAlike) license
- Pixie Cups © Bernard Spragg. NZ is licensed under a Public Domain license
- proliferation of ascogenous hyphae, ascus, Ascomycota © M. Piepenbring is licensed under a CC BY-SA (Attribution ShareAlike) license
- Ascomata © Medmyco is licensed under a CC0 (Creative Commons Zero) license
- Morel © Ejdzej is licensed under a CC BY-SA (Attribution ShareAlike) license
- Truffle © moi-même is licensed under a Public Domain license
- Dikaryon diagram © M. Piepenbring is licensed under a CC BY-SA (Attribution ShareAlike) license
- Basidium schematic © Debivort is licensed under a CC BY-SA (Attribution ShareAlike) license
- Fly agaric mushroom © Bernard Spragg. NZ is licensed under a Public Domain license
- Suillus bovinus © Algirdas is licensed under a Public Domain license
- Bird’s nest fungi © Phyzome is licensed under a CC BY-SA (Attribution ShareAlike) license
- Corn smut © Tomascastelazo is licensed under a CC BY-SA (Attribution ShareAlike) license
- Ustilago nuda at winterbarley © Rasbak is licensed under a CC BY-SA (Attribution ShareAlike) license
- Ruse on Rose © gailhampshire is licensed under a CC BY (Attribution) license
- Life cycle of Puccinia graminis © Agricultural Research Service is licensed under a Public Domain license