Bacteriological Culture Methods
The Birth of Bacteriology
While perhaps best known to us as a cause of human disease, bacteria really should be far more famous for their positive contributions than for their negative ones. Below, list three positive things that bacteria do for you.
- ______________________________________________________________________
- ______________________________________________________________________
- ______________________________________________________________________
Bacteria were first observed by Anton von Leeuwenhoek in the late 17th century, but didn’t become the objects of serious scientific study until the 19th century, when it became apparent that some species caused human diseases. The methods devised by Robert Koch, Louis Pasteur, and their associates during the “Golden Age” of microbiology, which spanned from the mid-1800s to early 1900s, are still widely used today. Most of these methods involved isolating single bacteria derived from a natural source (such as a diseased animal or human) and cultivating them in an artificial environment as a pure culture to facilitate additional studies.
During the middle of the twentieth century, when we believed we had defeated them at their disease-causing game, bacteria became popular subjects of empirical study in fields such as genetics, genetic engineering, and biochemistry. With the evolution of antibiotic-resistant strains and our increased knowledge of bacterial stealth attack strategies such as biofilms and intracellular growth, medical researchers have refocused their attention on disease-causing bacteria and are looking for new ways to defeat them.
Growing bacteria in pure culture is still one of the most widely used methods in microbiology. Many bacteria, particularly those that cause diseases and those used in scientific studies, are heterotrophic, which means that they rely on organic compounds as food, to provide energy and carbon. Some bacteria also require added nutritional components such as vitamins in their diet. An appropriate physical environment must be created, where important factors such as temperature, pH, and the concentration of atmospheric gases (particularly oxygen) are controlled and maintained.
The nutritional needs of bacteria can be met through specialized microbiological media that typically contain extracts of proteins (as a source of carbon and nitrogen), inorganic salts such as potassium phosphate or sodium sulfate, and in some cases, carbohydrates such as glucose or lactose. For fastidious bacteria (meaning, those that are picky eaters) vitamins and/or other growth factors must be added as well.
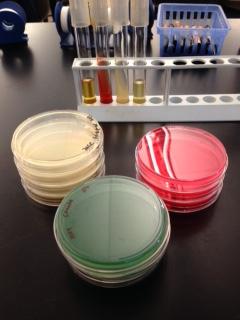
Bacteriological culture media can be prepared as a liquid (broth), a solid (plate media or slant media), or as a semi-solid (deeps) as illustrated in Figure 1. Solid and semi-solid media contain a solidifying agent such as agar or gelatin. Agar, which is a polysaccharide derived from red seaweed (Rhodophyceae) is preferred because it is an inert, non-nutritive substance. The agar provides a solid growth surface for the bacteria, upon which bacteria reproduce until the distinctive lumps of cells that we call colonies form.
Koch, Pasteur, and their colleagues in the 19th and early 20th centuries created media formulations that contained cow brains, potatoes, hay, and all sorts of other enticing microbial edibles. Today, bacteriological media formulations can be purchased in powdered form, so that all the preparer has to do is to measure out the correct amount, add the right amount of water, and mix. After the basic formula has been prepared, the medium is sterilized in an autoclave, which produces steam under pressure and achieves temperatures above boiling. Once sterilized media has cooled, it is ready to be used.
Growing Bacteria in Culture
A population of bacteria grown in the laboratory is referred to as a culture. A pure culture contains only one single type; a mixed culture contains two or more different bacteria. If a bacterial culture is left in the same media for too long, the cells use up the available nutrients, excrete toxic metabolites, and eventually the entire population will die. Thus bacterial cultures must be periodically transferred, or subcultured, to new media to keep the bacterial population growing.
Microbiologists use subculturing techniques to grow and maintain bacterial cultures, to examine cultures for purity or morphology, or to determine the number of viable organisms. In clinical laboratories, subculturing is used to obtain a pure culture of an infectious agent, and also for studies leading to the identification of the pathogen. Because bacteria can live almost anywhere, subculturing steps must be performed aseptically, to ensure that unwanted bacterial or fungal contamination is kept out of an important culture.
In microbiology, aseptic techniques essentially require only common sense and good laboratory skills. First, consider that every surface you touch and the air that you breathe may be contaminated by microorganisms. Then think about the steps you can take to minimize your exposure to unwanted invisible intruders. You should also be thinking about how to prevent contamination of your bacterial cultures with bacteria from the surrounding environment (which includes you).
To maintain an aseptic work environment, everything you work with should be initially free of microbes. Thus, we begin with pre-sterilized pipettes, culture tubes, and glassware. Inoculating loops and needles made of metal wire can be used to transfer bacteria from one medium to another, such as from the surface of an agar plate to a broth. Metal tools may be sterilized by heating them in the flame of a Bunsen burner. Glass tools or metal spreaders or forceps that can’t be sterilized by direct heat are dipped in alcohol followed by a brief pass through the flame to speed the evaporation process. Standard aseptic techniques used for culturing bacteria will be demonstrated at the beginning of lab.

One very important method in microbiology is to isolate a single type of bacteria from a source that contains many. The most effective way to do this is the streak plate method, which dilutes the individual cells by spreading them over the surface of an agar plate (see Figure 2). Single cells reproduce and create millions of clones, which all pile up on top of the original cell. The piles of bacterial cells observed after an incubation period are called colonies. Each colony represents the descendants of a single bacterial cell, and therefore, all of the cells in the colonies are clones. Therefore, when you transfer a single colony from the streak plate to new media, you have achieved a pure culture with only one type of bacteria.
Different bacteria give rise to colonies that may be quite distinct to the bacterial species that created it. Therefore, a useful preliminary step in identifying bacteria is to examine a characteristic called colonial morphology, which is defined as the appearance of the colonies on an agar plate or slant. Ideally, these determinations should be made by looking at a single colony; however, if the colonial growth is more abundant and single colonies are absent, it is still possible to describe some of the colonial characteristics, such as the texture and color of the bacterial growth.
Describing Colonial Morphology of Bacteria
By looking closely at the colonial growth on the surface of a solid medium, characteristics such as surface texture, transparency, and the color or hue of the growth can be described. The following three characteristics are readily apparent whether you’re looking at a single bacterial colony or more dense growth, without the aid of any type of magnifying device.
Texture—describes how the surface of the colony appears. Common terms used to describe texture may include smooth, glistening, mucoid, slimy, dry, powdery, flaky etc.
Transparency—colonies may be transparent (you can see through them), translucent (light passes through them), or opaque (solid-appearing).
Color or Pigmentation—many bacteria produce intracellular pigments which cause their colonies to appear a distinct color, such as yellow, pink, purple or red. Many bacteria do not produce any pigment and appear white or gray.
As the bacterial population increases in number, the colonies get larger and begin to take on a shape or form. These can be quite distinctive and provide a good way to tell colonies apart when they are similar in color or texture. The following three characteristics can be described for bacteria when a single, separate colony can be observed. It may be helpful to use a magnifying tool, such as a colony counter or dissecting microscope, to enable a close-up view of the colonies. Colonies should be described as to their overall size, their shape or form, what a close-up of the edges of the colony looks like (edge or margin of the colony), and how the colony appears when you observe it from the side (elevation).
Figure 4 shows a close-up of colonies growing on the surface of an agar plate. In this example, the differences between the two bacteria are obvious, because each has a distinctive colonial morphology.
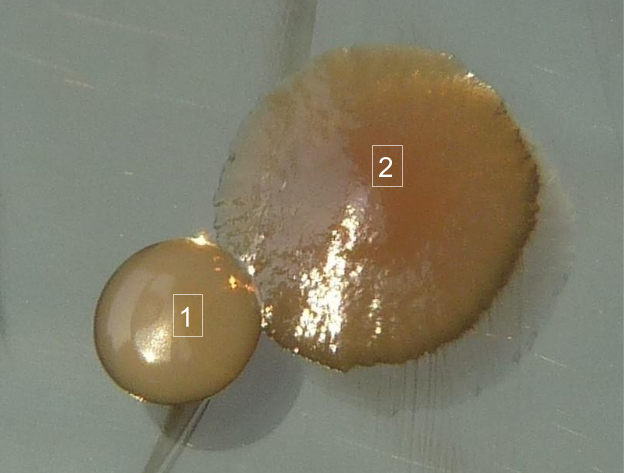
Using microbiology terms, describe fully the colonial morphology of the two colonies shown above. A full description will include texture, transparency, color, and form (size, overall shape, margin, and elevation).
| Colony 1 | |
| Colony 2 |
Size: ___________________________________________________________________
Texture: ________________________________________________________________
Transparency: ___________________________________________________________
Pigmentation: ___________________________________________________________
Form (shape, margin, elevation): ____________________________________________
Media Considerations
A culture medium must contain adequate nutrients to support bacterial growth. Minimally, this would include organic compounds that can provide the building blocks necessary for cellular reproduction. In many cases, predigested protein, such as hydrolyzed soy protein, serves this purpose and will support the growth of many different bacteria. These media formulations are generally referred to as complex media, to indicate that it is a mixture with many components.
Many media contain additional substances such as an antibiotic that may be selective for a particular type of bacteria by inhibiting most or all other types. Differential media will have additional compounds that permit us to distinguish among bacterial types based on differences in growth patterns. We will eventually use selective and differential media in our experiments, but the focus of this lab is to learn the basic culturing techniques, and therefore, the media used will be Tryptic Soy medium, a complex medium formulated with hydrolyzed soy protein.
The media you use in this lab and in all of the future labs will have already been prepared, but it is important for you as a budding microbiologist to understand and appreciate how culture media is prepared. With this in mind, your instructor may have you watch a brief video that demonstrates the art of media making.

Liquid media
Pre-sterilized glass or plastic graduated pipettes (Figure 5) are used to transfer specific volumes of sterile liquids accurately. It is important that you learn how to use these tools correctly, since it may be necessary to transfer sterile and sometimes contaminated liquids among various bottles and tubes. Their appropriate use will be discussed and demonstrated in lab. Some tips to remember:
- The pipette and the media are sterile; there should never be any direct contact with your hands, skin, or lab surfaces.
- Caps or lids on tubes or bottles should never be set down on lab surfaces.
- Tubes or bottles should be held at an angle during the transfer process, to minimize the potential for airborne contaminants to make their way into the opening.
- Passing the opening of the tube or bottle briefly through a flame before and after the transfer process will discourage airborne contaminants from getting into the sterile liquid.

Pipette practice:
- Obtain water in a small beaker, a 10 ml sterile graduated pipette, and a pipette aid (pipump). Take a minute to note the divisions on the pipette and to understand what volume each mark represents. Use of the pipette to transfer liquids will be demonstrated. Before trying to pipette a sterile liquid, practice drawing up 5 ml of water from the beaker, and releasing it back into the beaker in 1 ml increments. Continue until you feel comfortable holding the pipette and using the pipump. Then practice it again with water in a capped media bottle using aseptic techniques.
A portion of a 10 mL graduated pipette is shown in Figure 6. What is the volume of liquid in this pipette?
Volume:________________________________
Solid and semi-solid media
Growing cultures of bacteria on solid media (agar plate or slant) permits us to view and identify colonial characteristics, and also provides a way to separate bacteria in a mixed culture. Cultures grown on agar plates usually don’t survive for long, since Petri dish lids are not tight fitting and the media (and bacteria) dehydrate. Cultures grown on agar plates should always be handled “bottom-up” to prevent condensation—which often accumulates on the lid of the dish during incubation—from dripping down on the culture.
Bacteria may be grown in agar slant or stab media in tubes if the purpose is to maintain them in a longer term culture. Generally, bacteria grown on slants will remain viable for a few weeks to a few months, and sometimes longer if stored in a refrigerator.
In this laboratory, you will be introduced to aseptic techniques and basic lab skills needed to grow and maintain bacteria in culture. You will be applying these skills often, so mastery is important.
Method
A volunteer from your lab bench should obtain one of each of the following cultures:
- TSA streak plate culture of Micrococcus luteus and Enterococcus faecalis
What BSL containment level practices should be used?
M. luteus__________________
E. faecalis__________________
- A “mixed culture” in TSB that contains two different bacteria
Below, write the names of the two bacteria in the mixed culture and the appropriate BSL, as specified by your instructor:
Mixed culture bacterium 1 ____________________________________________________
Mixed culture bacterium 2 ____________________________________________________
The techniques needed will first be demonstrated by your instructor. After the demonstration, perform the following tasks, and record your observations/results.
Broth Subculture
Obtain 2 sterile glass culture tubes, a bottle of Tryptic Soy Broth (TSB) and a test tube rack. With small pieces of colored tape, label each tube with your name and either “S” for subculture, or “C” for control. Using aseptic technique, use a 10 ml graduated pipette to transfer 2 ml of broth to each tube.
As demonstrated, use a flame-sterilized inoculating loop to pick up from the surface of the M. luteus streak plate culture, a single colony (if small) or a part of a colony (if large) and transfer it to the broth in the tube labeled “S.” Add nothing to the second tube “C” which will serve as a sterility control. Note how the broths look immediately after you inoculate them (they should still look mostly clear). Bacterial growth in broths is indicated by the development of a cloudy appearance. If the newly inoculated broth looks cloudy at the start, you will have no way to determine if this is due to bacterial growth during the incubation period. If your broth looks cloudy, discard it and make another broth using less bacteria.
Place the broth subcultures in an incubator at the temperature and time specified by your instructor.
Streak Plate
Separation of a mixed culture into individual colonies that can be subcultured to make pure cultures depends on how well the streak plate is prepared. The goal of streak plate method is to dilute the cells by spreading them out over the surface of the agar. This is accomplished in stages, as will be demonstrated in lab before you try it yourself.
Use the simulated agar surface below to practice the streak pattern using a pen or pencil.

Obtain two TSA plates, and write your name on the bottom half (the half containing the media) around the edge and following the curve (so the writing won’t hide your view of the bacterial colonies once they grow). Also write M. luteus on one plate (the name of the bacteria you will subculture to this plate). On the other, write “mixed” to indicate that you’re subculturing from the mixed culture broth to this plate.
As demonstrated, use a sterilized inoculating loop to pick up one M. luteus colony (or a piece of a colony) and transfer it to the surface of the agar plate. Spread the bacteria over approximately a quarter of the plate, edge to edge. Consider this step 1.
Flame the loop and cool it in the agar. Overlap the step 1 streak 3-4 times to pull out a reduced number of bacteria, and spread them out down the side of the plate. Consider this step 2.
Flame the loop and cool it in the agar. Overlap the step 2 streak 3-4 times and spread over the surface. Continue this process, flaming the loop in between each step, until the entire surface of the agar plate is covered.
After performing this with the M. luteus culture for practice, repeat the process with a drop of the mixed culture broth that you transfer to the plate with a sterile inoculating loop.
Place the streak plate subcultures in an incubator at the temperature and time specified by your instructor.
Slant Subculture of M. luteus
Obtain one slant tube containing TSA, and label it using a small piece of tape with your name and culture name (M. luteus). Using a sterilized inoculating loop, pick up a bacterial colony (or piece of a colony) from the surface of the plate culture of M. luteus, and inoculate the surface of the slant. Place the slant subculture in an incubator at the temperature and time specified by your instructor.
Stab or Deep Tube Subculture of E. faecalis
Obtain one stab tube containing semisolid TSA, and label it using a small piece of tape with your name and culture name (E. faecalis). Using a sterilized inoculating needle, pick up a bacterial colony (or piece of a colony) from the surface of the plate culture of E. faecalis, and inoculate the media by stabbing the needle into the center of the agar in the tube,and pushing it down to the bottom. Withdraw the needle carefully and try to remove it by following the same stab line that you made pushing the needle down. Place the stab subculture in an incubator at the temperature and time specified by your instructor.
A note about incubation temperatures
As you will learn, bacteria have preferred growth temperatures where their reproduction rate is the greatest. All of the bacteria we work with in lab are mesophilic, which means that they grow at temperatures between 20–40°C. However, some prefer body temperature (37°C), while others grow best at room temperature (approximately 25°C). This lab is equipped with incubators set at either temperature.
How long you plan to leave your cultures in an incubator should also be a consideration. Growing cultures at the higher temperature may speed their rate of growth, but it also causes dehydration of the media and an earlier demise to the bacteria in the culture.
As a general rule, for bacteria that grow best at body temperature, if you intend on returning to lab within 24 to 36 hours (highly recommended), then incubate them at 37°C. If you cannot return to lab during an “open lab” period, then incubate them at room temperature, or arrange to have your cultures transferred to a refrigerator after they grow, so that the culture won’t die out before you can finish your experiments. Bacteria that grow best at room temperature should always be incubated at room temperature, and growth may take a little longer.
Primary culture from an environmental source—you!
With your introduction to basic bacteriological culturing techniques complete, it’s time to apply those skills. Today is the beginning of The Human Skin Microbiome Project, which starts with the primary culture of bacteria from your skin on TSA medium. It is important that you read the project description (in the next chapter) so that you understand the goals and the scope of the project.
To begin, you will take a sample from your skin. Your first decision will be what part of your skin do you want to sample? Note: ONLY external skin surfaces are permitted.
Obtain a sterile swab and a tube of sterile distilled water, and label a TSA plate with your name and the date. Remove the wrapping from the swab and soak it in sterile water, using aseptic technique.
Rub the wet swab back and forth firmly over the area of skin you have chosen to sample. Then rub the swab over approximately a third of the surface of the TSA agar plate.
Sterilize an inoculating loop, and complete the rest of the streak plate pattern using the loop. Incubate this plate at room temperature for up to a week.
After incubation, look to see if isolated colonies have developed on the plate. If there are no colonies or no isolated colonies, you will need to make another streak plate with the advice of your instructor on how to proceed. If there are isolated colonies, transfer the plate to the refrigerator. From this plate you will ultimately choose one single colony and prepare a pure culture. The criteria for colony selection and next steps are described in the next chapter, “The Human Skin Microbiome Project.”
To complete the lab, the bacteria in the cultures have to grow. Therefore, the following observations are made AFTER the cultures have had time to grow.
Observations and Outcomes
Broth subcultures
Look at the broth subculture tubes, and describe what you expected to see, and how they appear in terms of how “cloudy” they look—cloudiness is an indication of bacterial growth.
|
|
Cloudiness of broth before incubation | Predicted appearance of broth after incubation | Actual appearance of broth after incubation |
|
M. luteus subculture (“S”)
|
|||
|
Sterility Control (“C”)
|
Streak plate subcultures
Look at the streak plate subcultures that you made. Conduct a self-assessment of how well you performed the technique. What you hope to see are individual colonies, well separated from each other. On the streak plate of the mixed culture, you should be able to see two distinctly different types of colonies.
M. luteus streak plate:
- Are the colonies well separated?
- How many different types of colonies do you see?
- Describe in full the colonial morphology of the bacteria on this plate:
Streak plate of the mixed culture:
- Are the colonies well separated?
- Could you make a pure culture of both bacteria from this plate? If you think you can, subculture a single colony of each type to one half of a TSA plate, divided by drawing a line with a marker on the bottom of the plate, as shown below. Incubate the plate, and then observe to see if you successfully separated the two bacteria in the mixed culture into two pure cultures. Use this self-analysis to consider improvements you might make in the technique you applied to making the streak plate.
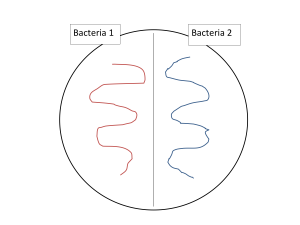
- Describe in full the colonial morphology of both of the bacteria from the mixed culture:
| Colony Type 1 | Colony Type 2 | |
| Size | ||
| Texture | ||
| Transparency | ||
| Pigmentation | ||
| Whole Colony |
TSA slant subculture
Examine the subculture of M. luteus you prepared on the TSA slant.
- Describe the texture, transparency, and pigmentation of the bacterial growth on the slant. Only these characteristics can be described for a slant culture, since there should be no discreet colonies on the slant, only an area of dense growth along the streak line.
- Does your description match what was noted for the M. luteus colonies when you described the colonial morphology previously?
- Do you see evidence of any other type of bacteria (meaning a different colonial morphology) on the slant?
- Is this a pure culture?
TSA stab subculture
Look closely along the stab line in the media in the tube. Do you see evidence of bacterial growth? If yes, describe and/or sketch how it appears.
Semisolid agar of the type used in this exercise can be used as a way to evaluate if a bacteria is motile, meaning in possession of one or more flagella that facilitates movement through liquids or semisolids. The way to evaluate motility is to look closely at the line of inoculation you created when the tube was stabbed. Nonmotile bacteria will grow along the stab line only. If they are motile, they will be able to move through the semisolid agar (like swimming through jello), and you won’t be able to see a distinct line in the agar—just cloudiness surrounding the stab line.
- Based on your observation of the bacteria in the stab culture, is there evidence that the bacteria are motile?
- For bacteria, the ability to move (motility) requires that they have which specific cellular structure?

